 
For older and/or smaller samples with very little carbon-14, a mass spectrometer can be used, distinguishing the charge and mass of different ions vaporized from the sample. And so forth.įor younger and/or larger samples that contain more carbon-14, the carbon-14 content can generally be measured by a radiation counter to directly detect beta decay of carbon-14 into nitrogen-14. After the organism has been dead for 17,190 years (5,730 x 3), it should contain 1/2 3 = 1/8 of the original amount of carbon-14. After the organism has been dead for 11,460 years (5,730 x 2), it should contain 1/2 2 = 1/4 of the original amount of carbon-14. After the organism has been dead for about 5,730 years, it should contain 1/2 1 = 1/2 of that original amount of carbon-14. Carbon-14, which has a half-life of approximately 5,730 years, decays at a rate of about 13.5 atoms/minute per gram of total carbon. As long as an organism is living and exchanging carbon with the environment, the percentage of carbon-14 in an organism should remain approximately equal to that of the environment. Approximately 1 atom out of every 10 12 newly acquired carbon atoms is carbon-14. Living organisms acquire carbon from atmospheric CO 2 either directly for plants or indirectly for other organisms (herbivorous animals eat plants that have acquired atmospheric carbon, and carnivorous animals eat herbivorous animals that have eaten plants that acquired atmospheric carbon). Roughly 1×10 -12 of the carbon in atmospheric CO 2 is carbon-14. If a neutron generated by a cosmic ray knocks a proton out of 14N and takes its place, carbon-14 (its nucleus contains 6 protons and 8 neutrons) is formed:Ĭarbon in the atmosphere combines with oxygen to form carbon dioxide or CO 2. Most of the atmosphere is made of 14N (its nucleus contains 7 protons and 7 neutrons). High-energy cosmic rays from the sun or other sources in space hit atoms in the upper atmosphere, knocking loose neutrons. How is carbon-14 formed in the atmosphere? The beta-minus decay equation for carbon-14 is: 14C → 14N + e – (and an anti-neutrino)ģ. Beta decay occurs when an atom trades a neutron for a proton or vice versa, in order to reach a more stable, lower-energy state. In beta-plus decay, a proton turns into a neutron plus a positron (and a neutrino). In beta-minus decay, a neutron turns into a positively charged proton plus an electron (along with a neutral particle called an anti-neutrino). What is beta decay? Write the decay equation for carbon-14.īeta particles are electrons (which have negative electric charge) and positrons (the positively charged antimatter version of an electron). Carbon-14 or 14C (its nucleus contains 6 protons and 8 neutrons) decays with a half-life of approximately 5,730 years to become nitrogen-14 or 14N (its nucleus contains 7 protons and 7 neutrons).Ģ. Carbon-11 or 11C (its nucleus contains 6 protons and 5 neutrons) decays with a half-life of approximately 20 minutes to become stable boron-11 or 11B (its nucleus contains 5 protons and 6 neutrons). If certain other elements are exposed to radiation, they can temporarily form other carbon isotopes that are radioactive. Both of those carbon isotopes are stable - they do not radioactively decay to become another element. Approximately 1 percent is carbon-13 or 13C (its nucleus contains 6 protons and 7 neutrons). 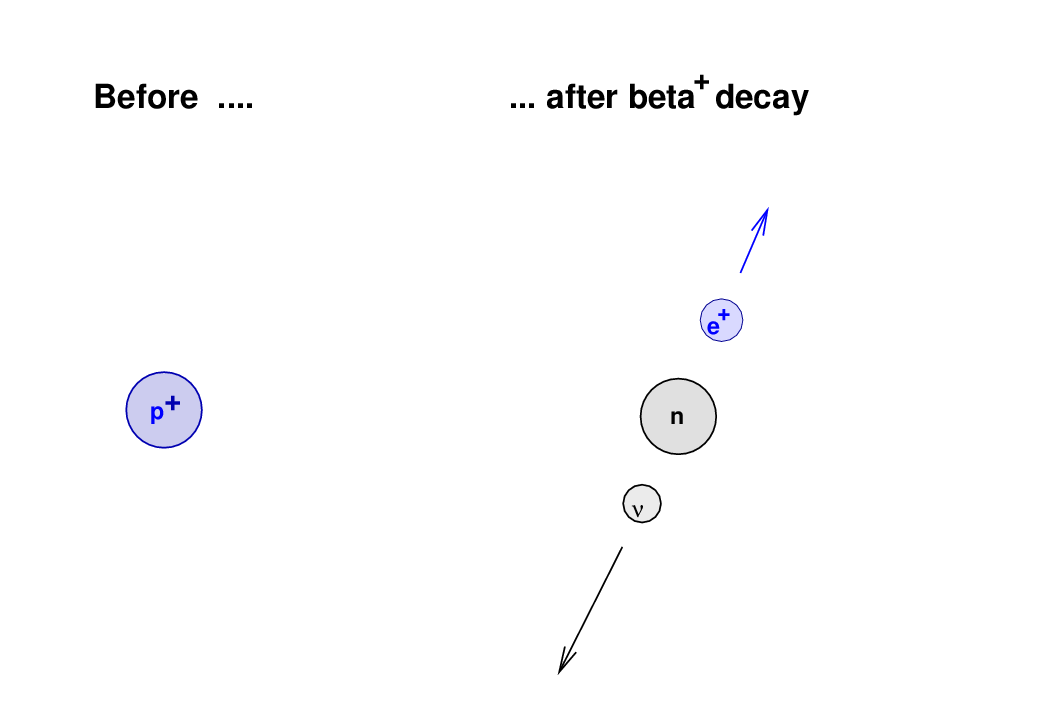
Most of the naturally occurring carbon on Earth is carbon-12 or 12C (its nucleus contains 6 protons and 6 neutrons). Isotopes are atoms of the same element with different masses - they have the same numbers of protons and same numbers of electrons, but different numbers of neutrons. What are isotopes, and what are the isotopes of carbon? What carbon isotopes are radioactive? 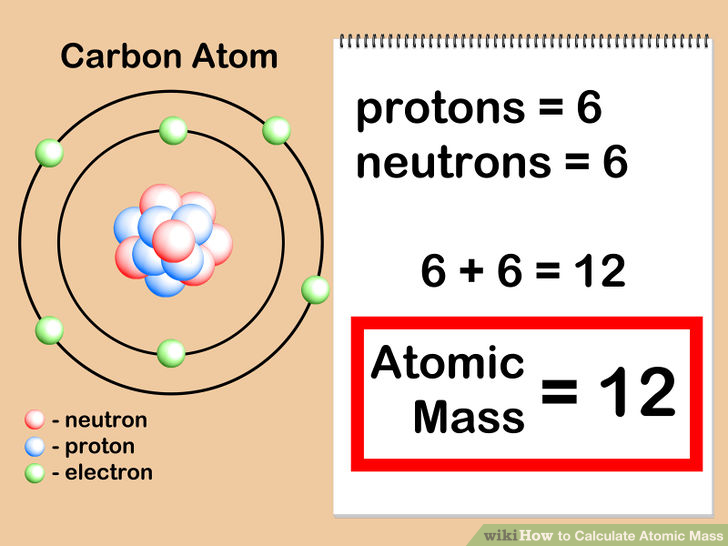
PHYSICAL AND CHEMICAL SCIENCES Discussion questions:ġ. Directions: After students have had a chance to review the article “ Mosses tell story of retreating ice,” lead a classroom discussion based on the questions that follow. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
